|
The combination of nivolumab (anti-PD1) and ipilimumab (anti-CTLA-4) has shown remarkable success in melanoma and renal cell carcinoma ( 10- 12). 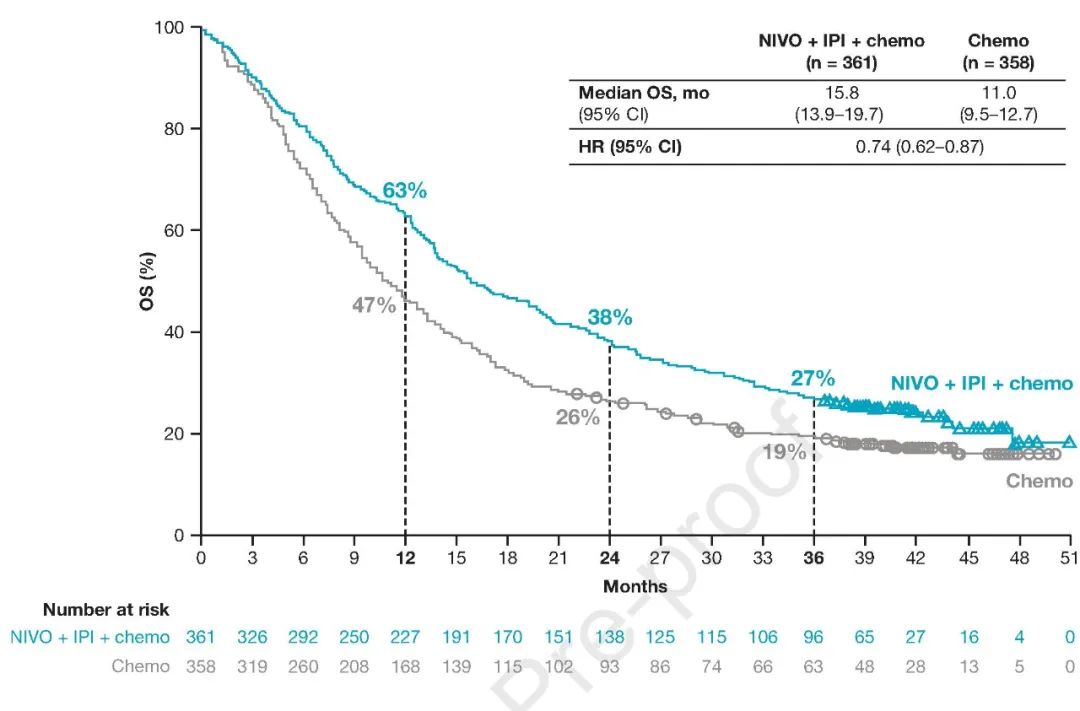
In that context there has been ongoing interest in the combination of PD1/PD-L1 inhibitors with the other established class of checkpoint inhibitors targeting CTLA-4. Multiple strategies have been employed to increase response rates, including combination with chemotherapy as well as other immune modulatory drugs. 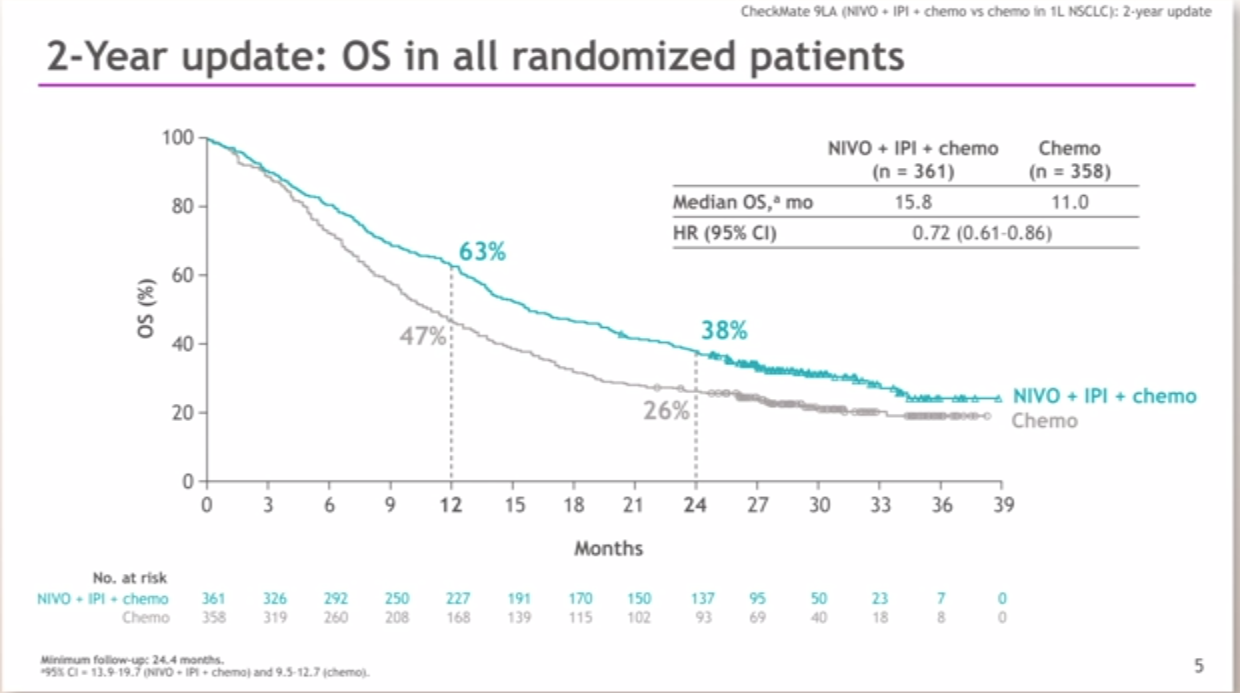
However, many patients, especially those with tumors expressing no or low PD-L1, do not respond to anti-PD1/PD-L1 drugs. Treatment with PD1/PD-L1 inhibitors has produced significant improvements in survival for patients when compared with chemotherapy, and a subset of patients experience remarkably prolonged remissions ( 5). Several landmark trials were published between 2015–2017 demonstrating the efficacy of drugs targeting the PD1/PD-L1 pathway in both the refractory and the first-line setting for mNSCLC ( 4- 9). Monoclonal antibodies targeting two of these pathways, PD1/PD-L1 and CTLA-4, have revolutionized the treatment of many types of cancer, including NSCLC. Drugs targeting these pathways “take the brakes” off the immune system, allowing the body’s own immune system to act. Increasing understanding of the importance of the immune system in cancer pathogenesis led to the discovery of “immune checkpoints”: these inhibitory pathways were shown to be upregulated in tumors and the surrounding microenvironment, suppressing the ability of the immune system to react to and destroy cancer cells ( 3). But perhaps the most significant progress in the treatment of NSCLC came with the introduction of checkpoint inhibitors into the cancer treatment landscape. 
As our understanding of cancer biology expanded during the 2000s–2010s, better drugs were added to our armamentarium with the introduction of targeted therapies for oncogene-addicted NSCLC and anti-VEGF drugs added to traditional chemotherapy. While these regimens improved survival, they did so at the cost of toxic side effects and long-term prognosis remained poor ( 2). Cytotoxic chemotherapy was the standard-and only-treatment for metastatic lung cancer until the mid-2000s, with numerous drugs and combinations trialed. While this remains a deadly disease, there has been a significant expansion of the number of treatment options available to patients with metastatic NSCLC (mNSCLC) over the last 10 years. Lung cancer is the second most common cancer in the United States and the leading cause of cancer death, with over 135,000 people expected to succumb by the end of 2020 ( 1). Email: 23 October 2020 Accepted: 16 November 2020 Published: 30 December 2020. Associate Professor of Internal Medicine (Medical Oncology), Yale School of Medicine/Yale Cancer Center, New Haven, CT, USA. Policy of Dealing with Allegations of Research MisconductĬorrespondence to: Sarah B.Policy of Screening for Plagiarism Process.Here we review phase 3 clinical trials utilizing immunotherapy in the first line for treatment of NSCLC, including Pembrolizumab in KEYNOTE-024, KEYNOTE-042, KEYNOTE-189 and KEYNOTE-407 Nivolumab and Ipilimumab in CHECKMATE-227 and CHECKMATE 9LA and Atezolizumab in IMpower110, IMpower130 and IMpower150.ĬTLA-4 chemotherapy immune checkpoints inhibitors lung cancer monoclonal recombinant antibodies programmed death receptor. 
Patients with PD-L1 1% immunotherapy doublet, Nivolumab and Ipilimumab, or single agent immunotherapy combined with chemotherapy. Patients with tumor expression levels of PD-L1 ≥ 50% are candidates for treatment with single agent Pembrolizumab or Atezolizumab. The inclusion criteria differ between the various clinical trials, including the cut-off levels of PD-L1 expression on tumor cells, and the tumor histology (squamous or non-squamous). Multiple phase 3 studies have tested different medications targeting programmed death receptor 1 (PD-1), programmed death-ligand 1 (PD-L1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), with or without chemotherapy. Immunotherapy for non-small cell lung cancer (NSCLC) is incorporated increasingly in first line treatments protocols.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
